|
2/26/2023 0 Comments O3 molecular geometry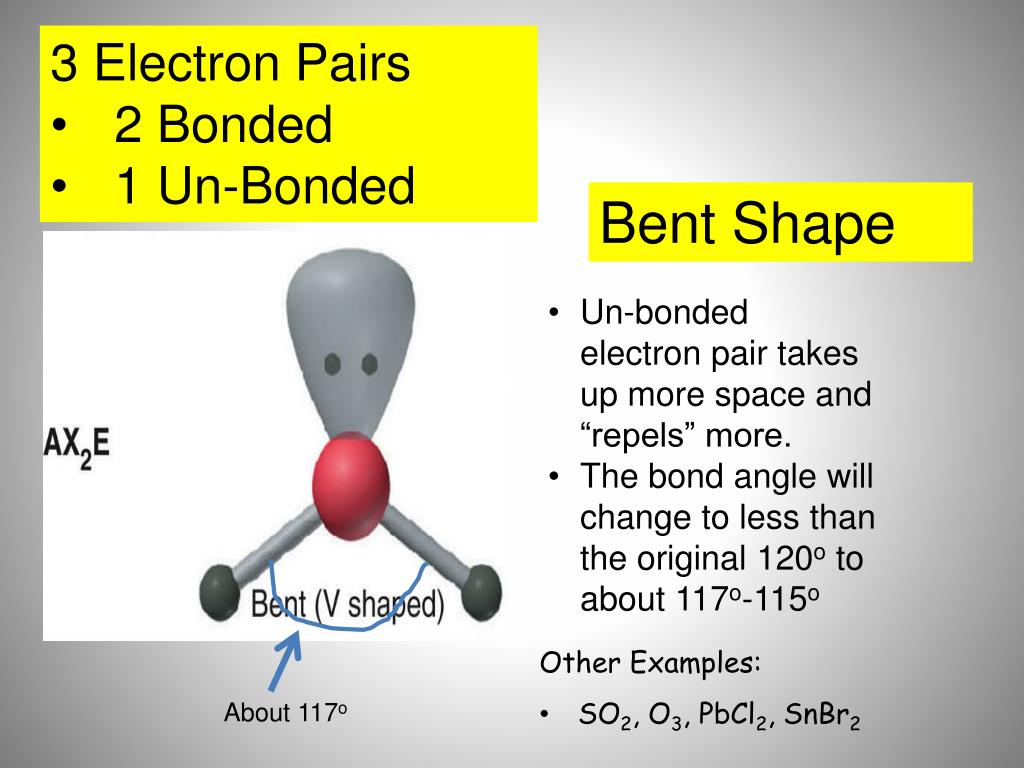
It is important to know the Lewis structure of a molecule to understand its physical properties, hybridization, and shape of the molecule. Molecules have polarity because it has both the charges that are a partially positively charged end and the partially negatively charged end. Our videos prepare you to succeed in your college classes. 
Answer = C2Cl4 ( Tetrachloroethylene ) is nonPolar What is polar and non-polar?, Using Standard Molar Entropies), Gibbs Free Energy Concepts and Calculations, Environment, Fossil Fuels, Alternative Fuels, Biological Examples (*DNA Structural Transitions, etc. Is CO Polar or Nonpolar? Question: Is H2SO3 an ionic or Molecular bond ? Tags: Question 6. In O3, the electric dipole moments of the bonds don’t counterbalance one another which results in a net dipole moment. So now the valence electrons on the central atom of iodine will be six. The reason the molecule is polar lies in the bonding between the three Oxygen atoms concerned. Clutch Prep is not sponsored or endorsed by any college or university. Due to this, O3 (Ozone) is polar in nature. In the O2 molecule, 2 oxygen atoms are joined together with a double bond between them. Pure ozone exists in a gaseous form of blue color with a heavy irritating odor. 
As per this rule, every atom should have eight electrons in their outer orbit. Answer = AsH3 ( Arsine ) is Polar What is polar and non-polar? SO2(Sulfur Dioxide) Molecular Geometry & Lewis Structure, CCL4 Molecular Geometry, Lewis Structure, Hybridization, And Everything, H2O Molecular Geometry, Lewis Structure, Shape and Bond Angles, Sf2 Molecular Geometry, Lewis Structure, Polarity and Bond Angles, HCN Lewis Structure, Molecular Geometry, Shape, and Polarity. There three molecules of iodine in this molecule and hence the name of the compound are Triodide. A polar molecule has a net dipole as a result of the opposing charges (i.e. for each compound please say if it is polar or not, what the molecular geometry is and explain the lewis dot for each please : NH2Cl, Cl2O, AsH5, AlH3, CCl2F2, O3 On liquidation of ozone, it changes to a deep blue colored liquid. This bent shape results in net electric dipole which imparts polar nature in the O3 molecule. The polarity and non-polarity of a molecule can also be determined by just viewing the structural geometry of a molecule. Question = Is O3 polar or nonpolar ? Answer: O3 is a polar molecule due to the presence of lone pair electrons on the central oxygen atom. 11616 views Draw the Lewis structure, then determine the shape of … As it forms the pair of electrons now, there will be three lone pairs, and two bond pairs of the electrons as each Iodine atoms has a bond with the central atom sharing one electron each in total there 3 lone pairs and 2 bond pairs on the central atom. Hence, by virtue of covalent bond and zero permanent dipole moment, the diatomic oxygen molecule (O2) is non-polar in nature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
